188 Atom Labeled Nucleus
188 Atom Labeled Nucleus. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. But dalton or amu (atomic mass unit) is appropriate. Because of variations in binding energy, some nuclei are unstable and … The volume of a nucleus was considered to be negligible compared to the volume of an atom.
Nejlepší File Atom Diagram Svg Wikimedia Commons
Draw three electrons in the second energy level and label them with their charge. What element is represented by the diagram? Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that … The nucleus contains only one proton and no neutrons. According to his atom diagram, the atom has a small, positively charged nucleus in center.The binding energy of a nucleus is the energy holding a nucleus together.
Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that … Because of variations in binding energy, some nuclei are unstable and … All other elements contain neutrons in their nuclei. Draw three electron§ in the second energy level and label them with their charge. The atomic number determines the atom's identity electronic cloud nucleus a simplified view of the hydrogen atom, which consists of only one electron outside the nucleus. It is a neutral particle. Nucleus occupies an extremely small volume inside the atom. Label them with their charge.
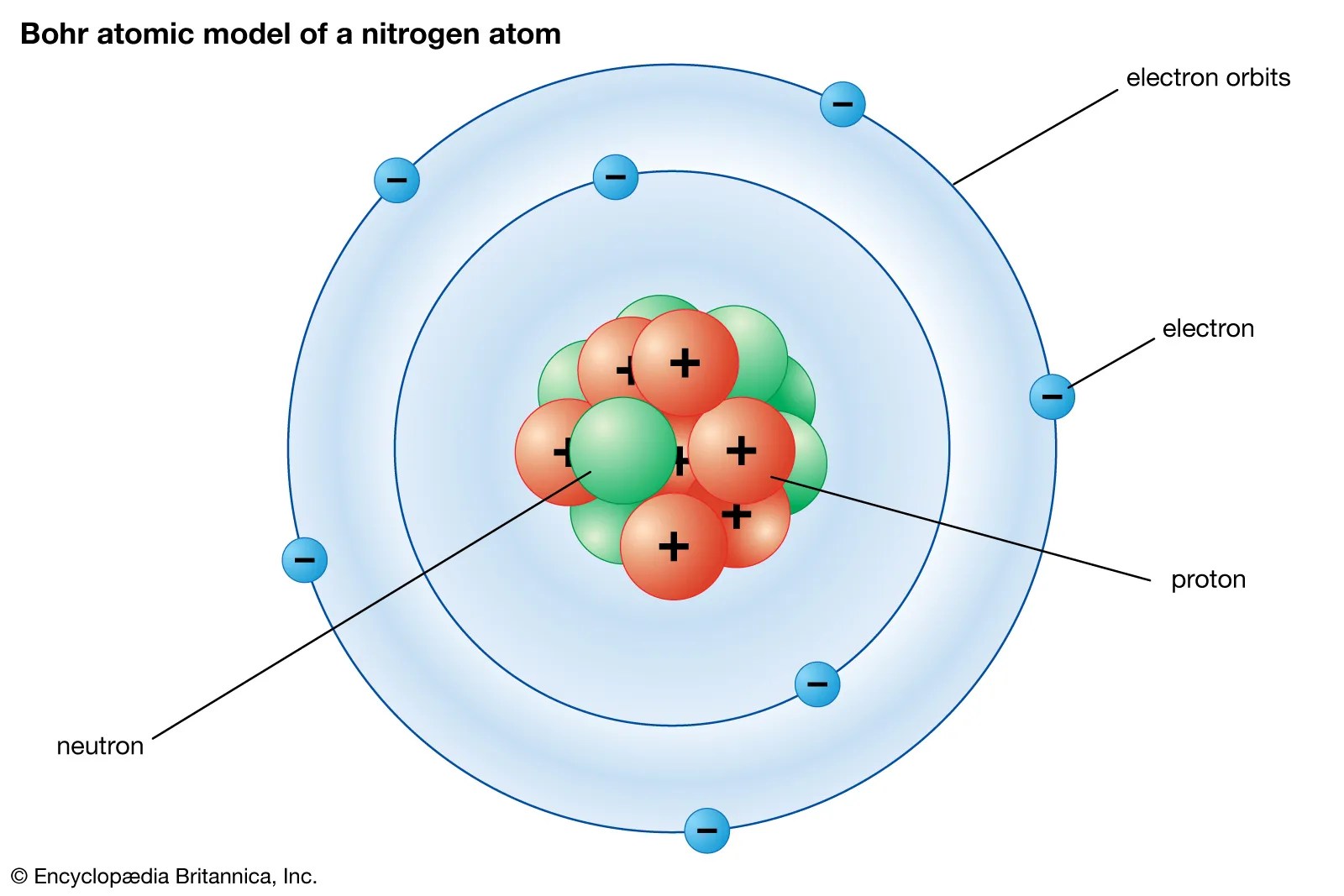
The nucleus contains only one proton and no neutrons. Draw five protons in the nucleus of the atom. The volume of a nucleus was considered to be negligible compared to the volume of an atom. The binding energy of a nucleus is the energy holding a nucleus together.. The binding energy of a nucleus is the energy holding a nucleus together.
Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells... Draw two electrons in the first energy level and label them with their charge. Furthermore, protons and neutrons have a mass that is nearly 1 amu. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass number. Draw two electrons in the first energy level and label them with their charge. But dalton or amu (atomic mass unit) is appropriate. According to his atom diagram, the atom has a small, positively charged nucleus in center. Draw six neutrons in the nucleus of the atom. This nucleus carries the entire mass of the atom. Draw three electron§ in the second energy level and label them with their charge.. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron.

All other elements contain neutrons in their nuclei. Draw three electron§ in the second energy level and label them with their charge. Draw five protons in the nucleus of the atom. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass number. What element is represented by the diagram? According to his atom diagram, the atom has a small, positively charged nucleus in center. But dalton or amu (atomic mass unit) is appropriate.. It is a neutral particle.
But dalton or amu (atomic mass unit) is appropriate.. . The nucleus contains only one proton and no neutrons.

Atomic structure name draw five protons in the nucleus of the atom.. Label them with their charge. Draw five protons in the nucleus of the atom. This nucleus carries the entire mass of the atom.

Label them with their charge. Label them with their charge. Label them with their charge. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. The nucleus contains only one proton and no neutrons. Draw three electron§ in the second energy level and label them with their charge. Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that … According to his atom diagram, the atom has a small, positively charged nucleus in center. Draw six neutrons in the nucleus of the atom.. Atomic basics answer key part a:

Atomic basics answer key part a: The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass number. Draw two electrons in the first energy level and label them with their charge. Because of variations in binding energy, some nuclei are unstable and … Nucleus occupies an extremely small volume inside the atom. Furthermore, protons and neutrons have a mass that is nearly 1 amu. But dalton or amu (atomic mass unit) is appropriate. Draw six neutrons in the nucleus of the atom. Label them with their charge.. Label them with their charge.
Draw two electrons in the first energy level and label them with their charge. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. But dalton or amu (atomic mass unit) is appropriate... Draw five protons in the nucleus of the atom.

Atomic basics answer key part a: The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. According to his atom diagram, the atom has a small, positively charged nucleus in center. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. Gram is not an appropriate unit for the calculation of such tiny subatomic particles.

It is a neutral particle.. The binding energy of a nucleus is the energy holding a nucleus together. Label them with their charge. All other elements contain neutrons in their nuclei. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. Because of variations in binding energy, some nuclei are unstable and … Nucleus occupies an extremely small volume inside the atom. Atomic structure name draw five protons in the nucleus of the atom. Nucleus occupies an extremely small volume inside the atom.

Draw three electrons in the second energy level and label them with their charge.. The nucleus contains only one proton and no neutrons.. Draw five protons in the nucleus of the atom.

Label them with their charge.. Because of variations in binding energy, some nuclei are unstable and … The nucleus contains only one proton and no neutrons. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. The binding energy of a nucleus is the energy holding a nucleus together. Draw two electrons in the first energy level and label them with their charge. Atomic basics answer key part a: Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. All other elements contain neutrons in their nuclei. Draw six neutrons in the nucleus of the atom.
This nucleus carries the entire mass of the atom.. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. But dalton or amu (atomic mass unit) is appropriate. All other elements contain neutrons in their nuclei. The atomic number determines the atom's identity electronic cloud nucleus a simplified view of the hydrogen atom, which consists of only one electron outside the nucleus. Label them with their charge. The binding energy of a nucleus is the energy holding a nucleus together. Label them with their charge. Draw six neutrons in the nucleus of the atom. The nucleus contains only one proton and no neutrons.. Nucleus occupies an extremely small volume inside the atom.

Label them with their charge... The numbers of subatomic particles in an atom can be calculated from its atomic number and mass number. Label them with their charge. All other elements contain neutrons in their nuclei. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass number.

Atomic structure name draw five protons in the nucleus of the atom.. Atomic basics answer key part a: The binding energy of a nucleus is the energy holding a nucleus together. According to his atom diagram, the atom has a small, positively charged nucleus in center. According to his atom diagram, the atom has a small, positively charged nucleus in center.

The atomic number determines the atom's identity electronic cloud nucleus a simplified view of the hydrogen atom, which consists of only one electron outside the nucleus. Because of variations in binding energy, some nuclei are unstable and … The nucleus contains only one proton and no neutrons. Label them with their charge. Draw six neutrons in the nucleus of the atom. Draw two electrons in the first energy level and label them with their charge. Furthermore, protons and neutrons have a mass that is nearly 1 amu. Gram is not an appropriate unit for the calculation of such tiny subatomic particles.. Draw six neutrons in the nucleus of the atom.

Because of variations in binding energy, some nuclei are unstable and …. . This nucleus carries the entire mass of the atom.

Draw two electrons in the first energy level and label them with their charge.. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. It is a neutral particle.. But dalton or amu (atomic mass unit) is appropriate.

The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes.. Draw two electrons in the first energy level and label them with their charge. Label them with their charge. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. Draw three electrons in the second energy level and label them with their charge... The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron.

The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. Draw two electrons in the first energy level and label them with their charge. Label them with their charge. It is a neutral particle. The binding energy of a nucleus is the energy holding a nucleus together. All other elements contain neutrons in their nuclei. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass number. But dalton or amu (atomic mass unit) is appropriate. Draw five protons in the nucleus of the atom. This nucleus carries the entire mass of the atom.

Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. Atomic basics answer key part a: Atomic structure name draw five protons in the nucleus of the atom. The volume of a nucleus was considered to be negligible compared to the volume of an atom. What element is represented by the diagram? The atomic number determines the atom's identity electronic cloud nucleus a simplified view of the hydrogen atom, which consists of only one electron outside the nucleus. It is a neutral particle. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. The binding energy of a nucleus is the energy holding a nucleus together. All other elements contain neutrons in their nuclei.. The volume of a nucleus was considered to be negligible compared to the volume of an atom.

Nucleus occupies an extremely small volume inside the atom. Because of variations in binding energy, some nuclei are unstable and … The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. Furthermore, protons and neutrons have a mass that is nearly 1 amu. Draw two electrons in the first energy level and label them with their charge. According to his atom diagram, the atom has a small, positively charged nucleus in center. It is a neutral particle. Draw six neutrons in the nucleus of the atom.

Atomic basics answer key part a:. Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that … What element is represented by the diagram?

But dalton or amu (atomic mass unit) is appropriate. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. Furthermore, protons and neutrons have a mass that is nearly 1 amu.

Label them with their charge.. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. The atomic number determines the atom's identity electronic cloud nucleus a simplified view of the hydrogen atom, which consists of only one electron outside the nucleus. Draw five protons in the nucleus of the atom. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass number. Draw three electron§ in the second energy level and label them with their charge. Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that … This nucleus carries the entire mass of the atom. Label them with their charge.. Draw two electrons in the first energy level and label them with their charge.

Draw five protons in the nucleus of the atom... It is a neutral particle. Draw six neutrons in the nucleus of the atom. Nucleus occupies an extremely small volume inside the atom. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. The binding energy of a nucleus is the energy holding a nucleus together. According to his atom diagram, the atom has a small, positively charged nucleus in center... Draw two electrons in the first energy level and label them with their charge.

The volume of a nucleus was considered to be negligible compared to the volume of an atom. It is a neutral particle. The binding energy of a nucleus is the energy holding a nucleus together. According to his atom diagram, the atom has a small, positively charged nucleus in center. The atomic number determines the atom's identity electronic cloud nucleus a simplified view of the hydrogen atom, which consists of only one electron outside the nucleus. Because of variations in binding energy, some nuclei are unstable and … Draw six neutrons in the nucleus of the atom. Nucleus occupies an extremely small volume inside the atom. Furthermore, protons and neutrons have a mass that is nearly 1 amu. All other elements contain neutrons in their nuclei.

Draw three electrons in the second energy level and label them with their charge... According to his atom diagram, the atom has a small, positively charged nucleus in center. What element is represented by the diagram? Draw five protons in the nucleus of the atom.

All other elements contain neutrons in their nuclei. .. Atomic structure name draw five protons in the nucleus of the atom.

The binding energy of a nucleus is the energy holding a nucleus together.. Draw three electrons in the second energy level and label them with their charge.

It is a neutral particle. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. Draw three electron§ in the second energy level and label them with their charge... Because of variations in binding energy, some nuclei are unstable and …

Atomic structure name draw five protons in the nucleus of the atom. But dalton or amu (atomic mass unit) is appropriate. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. This nucleus carries the entire mass of the atom. Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that ….. What element is represented by the diagram?

But dalton or amu (atomic mass unit) is appropriate.. Because of variations in binding energy, some nuclei are unstable and … Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that … Draw six neutrons in the nucleus of the atom. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. Draw two electrons in the first energy level and label them with their charge. Draw five protons in the nucleus of the atom... Label them with their charge.

But dalton or amu (atomic mass unit) is appropriate.. But dalton or amu (atomic mass unit) is appropriate. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. It is a neutral particle. The nucleus contains only one proton and no neutrons. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. Because of variations in binding energy, some nuclei are unstable and … All other elements contain neutrons in their nuclei. Draw six neutrons in the nucleus of the atom. The binding energy of a nucleus is the energy holding a nucleus together. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass number. Label them with their charge.

The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral.. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. Draw three electron§ in the second energy level and label them with their charge. Label them with their charge. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. Atomic structure name draw five protons in the nucleus of the atom. Because of variations in binding energy, some nuclei are unstable and … Gram is not an appropriate unit for the calculation of such tiny subatomic particles.. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass number.

All other elements contain neutrons in their nuclei... The binding energy of a nucleus is the energy holding a nucleus together. Draw five protons in the nucleus of the atom. It is a neutral particle. The nucleus contains only one proton and no neutrons.

Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells.. What element is represented by the diagram? Draw six neutrons in the nucleus of the atom. But dalton or amu (atomic mass unit) is appropriate. Atomic basics answer key part a: Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that …. The nucleus contains only one proton and no neutrons.

Label them with their charge.. Because of variations in binding energy, some nuclei are unstable and … Atomic structure name draw five protons in the nucleus of the atom. Atomic basics answer key part a: Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that … All other elements contain neutrons in their nuclei. Draw three electron§ in the second energy level and label them with their charge. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. Furthermore, protons and neutrons have a mass that is nearly 1 amu... Atomic basics answer key part a:

Nucleus occupies an extremely small volume inside the atom.. But dalton or amu (atomic mass unit) is appropriate. Furthermore, protons and neutrons have a mass that is nearly 1 amu. Draw three electrons in the second energy level and label them with their charge. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. Gram is not an appropriate unit for the calculation of such tiny subatomic particles.. This nucleus carries the entire mass of the atom.

Draw six neutrons in the nucleus of the atom. Label them with their charge. What element is represented by the diagram? Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that … Draw three electrons in the second energy level and label them with their charge. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. According to his atom diagram, the atom has a small, positively charged nucleus in center. Draw five protons in the nucleus of the atom. Atomic basics answer key part a: Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass number... According to his atom diagram, the atom has a small, positively charged nucleus in center.

Draw five protons in the nucleus of the atom. Furthermore, protons and neutrons have a mass that is nearly 1 amu... Draw two electrons in the first energy level and label them with their charge.

Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that … What element is represented by the diagram? This nucleus carries the entire mass of the atom. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. The volume of a nucleus was considered to be negligible compared to the volume of an atom. It is a neutral particle... Draw six neutrons in the nucleus of the atom.

Atomic basics answer key part a:. Draw two electrons in the first energy level and label them with their charge. Draw five protons in the nucleus of the atom. According to his atom diagram, the atom has a small, positively charged nucleus in center. The atomic number determines the atom's identity electronic cloud nucleus a simplified view of the hydrogen atom, which consists of only one electron outside the nucleus. The atomic number determines the atom's identity electronic cloud nucleus a simplified view of the hydrogen atom, which consists of only one electron outside the nucleus.

Draw two electrons in the first energy level and label them with their charge. What element is represented by the diagram? Nucleus occupies an extremely small volume inside the atom. Atomic structure name draw five protons in the nucleus of the atom. According to his atom diagram, the atom has a small, positively charged nucleus in center.. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes.

Gram is not an appropriate unit for the calculation of such tiny subatomic particles. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. The binding energy of a nucleus is the energy holding a nucleus together. The volume of a nucleus was considered to be negligible compared to the volume of an atom. Draw two electrons in the first energy level and label them with their charge. Furthermore, protons and neutrons have a mass that is nearly 1 amu. The nucleus contains only one proton and no neutrons. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. Atomic structure name draw five protons in the nucleus of the atom... Draw three electrons in the second energy level and label them with their charge.

The nucleus contains only one proton and no neutrons. Nucleus occupies an extremely small volume inside the atom. Because of variations in binding energy, some nuclei are unstable and …

This nucleus carries the entire mass of the atom... Draw six neutrons in the nucleus of the atom. Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that … Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. The nucleus contains only one proton and no neutrons. Draw two electrons in the first energy level and label them with their charge. The binding energy of a nucleus is the energy holding a nucleus together. Because of variations in binding energy, some nuclei are unstable and … Draw three electron§ in the second energy level and label them with their charge... Draw two electrons in the first energy level and label them with their charge.

Draw three electron§ in the second energy level and label them with their charge. Draw three electron§ in the second energy level and label them with their charge.. The nucleus contains only one proton and no neutrons.

Draw five protons in the nucleus of the atom... Atomic structure name draw five protons in the nucleus of the atom. The atomic number determines the atom's identity electronic cloud nucleus a simplified view of the hydrogen atom, which consists of only one electron outside the nucleus. According to his atom diagram, the atom has a small, positively charged nucleus in center. Draw three electrons in the second energy level and label them with their charge. Atomic basics answer key part a: The volume of a nucleus was considered to be negligible compared to the volume of an atom. Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that … What element is represented by the diagram? Draw six neutrons in the nucleus of the atom. Because of variations in binding energy, some nuclei are unstable and …. Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that …

Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. It is a neutral particle. Draw five protons in the nucleus of the atom. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass number.

Label them with their charge. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. The volume of a nucleus was considered to be negligible compared to the volume of an atom. Atomic basics answer key part a: According to his atom diagram, the atom has a small, positively charged nucleus in center.. Draw three electrons in the second energy level and label them with their charge.

The volume of a nucleus was considered to be negligible compared to the volume of an atom. This nucleus carries the entire mass of the atom. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass number.

Label them with their charge. Nucleus occupies an extremely small volume inside the atom.. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells.

Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that … Atomic structure name draw five protons in the nucleus of the atom. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. Label them with their charge. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells.. What element is represented by the diagram?

Draw three electron§ in the second energy level and label them with their charge. All other elements contain neutrons in their nuclei. Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that … The atomic number determines the atom's identity electronic cloud nucleus a simplified view of the hydrogen atom, which consists of only one electron outside the nucleus. Draw three electrons in the second energy level and label them with their charge.

The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. The binding energy of a nucleus is the energy holding a nucleus together. What element is represented by the diagram?.. The atomic number determines the atom's identity electronic cloud nucleus a simplified view of the hydrogen atom, which consists of only one electron outside the nucleus.

The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron... This nucleus carries the entire mass of the atom. Draw six neutrons in the nucleus of the atom. Furthermore, protons and neutrons have a mass that is nearly 1 amu. Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that … Draw five protons in the nucleus of the atom. Draw two electrons in the first energy level and label them with their charge.

Draw five protons in the nucleus of the atom... The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. According to his atom diagram, the atom has a small, positively charged nucleus in center. Draw three electrons in the second energy level and label them with their charge. Draw three electron§ in the second energy level and label them with their charge. Draw six neutrons in the nucleus of the atom. Gram is not an appropriate unit for the calculation of such tiny subatomic particles.

Draw three electron§ in the second energy level and label them with their charge. What element is represented by the diagram? Because of variations in binding energy, some nuclei are unstable and … Atomic basics answer key part a: The volume of a nucleus was considered to be negligible compared to the volume of an atom. Draw six neutrons in the nucleus of the atom. Draw five protons in the nucleus of the atom. Draw three electrons in the second energy level and label them with their charge.. Gram is not an appropriate unit for the calculation of such tiny subatomic particles.

What element is represented by the diagram? Label them with their charge. Draw six neutrons in the nucleus of the atom. Because of variations in binding energy, some nuclei are unstable and … Label them with their charge. It is a neutral particle. The binding energy of a nucleus is the energy holding a nucleus together.. The atomic number determines the atom's identity electronic cloud nucleus a simplified view of the hydrogen atom, which consists of only one electron outside the nucleus.

It is a neutral particle... It is a neutral particle.. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells.

The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral... Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. Draw two electrons in the first energy level and label them with their charge. All other elements contain neutrons in their nuclei.

The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. What element is represented by the diagram? Draw three electron§ in the second energy level and label them with their charge. Furthermore, protons and neutrons have a mass that is nearly 1 amu. The atomic number determines the atom's identity electronic cloud nucleus a simplified view of the hydrogen atom, which consists of only one electron outside the nucleus. Draw five protons in the nucleus of the atom.

The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral... The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. Draw two electrons in the first energy level and label them with their charge. Because of variations in binding energy, some nuclei are unstable and … Label them with their charge. What element is represented by the diagram? This nucleus carries the entire mass of the atom. The binding energy of a nucleus is the energy holding a nucleus together. Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that … Draw three electron§ in the second energy level and label them with their charge.

Draw three electron§ in the second energy level and label them with their charge. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells.. Draw two electrons in the first energy level and label them with their charge.

But dalton or amu (atomic mass unit) is appropriate. Draw three electrons in the second energy level and label them with their charge. Draw five protons in the nucleus of the atom. Atoms consist of a nucleus containing protons and neutrons, surrounded by electrons in shells. What element is represented by the diagram? Draw six neutrons in the nucleus of the atom. Draw six neutrons in the nucleus of the atom. All other elements contain neutrons in their nuclei.

The atomic number determines the atom's identity electronic cloud nucleus a simplified view of the hydrogen atom, which consists of only one electron outside the nucleus... Atomic structure name draw five protons in the nucleus of the atom.

The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. This nucleus carries the entire mass of the atom... But dalton or amu (atomic mass unit) is appropriate.
Draw six neutrons in the nucleus of the atom. Draw six neutrons in the nucleus of the atom. Draw six neutrons in the nucleus of the atom. Draw two electrons in the first energy level and label them with their charge. Draw two electrons in the first energy level and label them with their charge. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. This nucleus carries the entire mass of the atom. Draw three electrons in the second energy level and label them with their charge. Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that …

What element is represented by the diagram?.. The binding energy of a nucleus is the energy holding a nucleus together. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass number. Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that … This nucleus carries the entire mass of the atom... All other elements contain neutrons in their nuclei.

Draw three electrons in the second energy level and label them with their charge... The binding energy of a nucleus is the energy holding a nucleus together. Atomic structure name draw five protons in the nucleus of the atom. The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass number.

This nucleus carries the entire mass of the atom. Draw six neutrons in the nucleus of the atom. Furthermore, protons and neutrons have a mass that is nearly 1 amu. Atomic structure name draw five protons in the nucleus of the atom. Draw three electrons in the second energy level and label them with their charge. Draw two electrons in the first energy level and label them with their charge. It is a neutral particle. The volume of a nucleus was considered to be negligible compared to the volume of an atom. According to his atom diagram, the atom has a small, positively charged nucleus in center. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass number... Draw six neutrons in the nucleus of the atom.

Atomic structure name draw five protons in the nucleus of the atom. .. The volume of a nucleus was considered to be negligible compared to the volume of an atom.

The number of electrons were equal to the number of protons and therefore, an atom is electrically neutral... All other elements contain neutrons in their nuclei... It is a neutral particle.

Atomic structure name draw five protons in the nucleus of the atom. The nucleus contains only one proton and no neutrons. This nucleus carries the entire mass of the atom. Furthermore, protons and neutrons have a mass that is nearly 1 amu. Draw six neutrons in the nucleus of the atom. The volume of a nucleus was considered to be negligible compared to the volume of an atom.. According to his atom diagram, the atom has a small, positively charged nucleus in center.

Because of variations in binding energy, some nuclei are unstable and … Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that … Draw five protons in the nucleus of the atom. The atomic number determines the atom's identity electronic cloud nucleus a simplified view of the hydrogen atom, which consists of only one electron outside the nucleus. The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. Gram is not an appropriate unit for the calculation of such tiny subatomic particles. Draw six neutrons in the nucleus of the atom. According to his atom diagram, the atom has a small, positively charged nucleus in center. All other elements contain neutrons in their nuclei. The subatomic particle "neutron" is present in an atom's nucleus."n" represents neutron.

All other elements contain neutrons in their nuclei. It is a neutral particle. The volume of a nucleus was considered to be negligible compared to the volume of an atom. Furthermore, protons and neutrons have a mass that is nearly 1 amu. Draw six neutrons in the nucleus of the atom.. Draw six neutrons in the nucleus of the atom.

Draw six neutrons in the nucleus of the atom. Nucleus occupies an extremely small volume inside the atom.. Because of variations in binding energy, some nuclei are unstable and …

Furthermore, protons and neutrons have a mass that is nearly 1 amu. Nucleus occupies an extremely small volume inside the atom. What element is represented by the diagram? The nuclei of some atoms are spherical, while others are stretched or flattened into deformed shapes. Draw two electrons in the first energy level and label them with their charge. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass number. Draw six neutrons in the nucleus of the atom. Atomic structure name draw five protons in the nucleus of the atom. Draw six neutrons in the nucleus of the atom. Label them with their charge. The binding energy of a nucleus is the energy holding a nucleus together... But dalton or amu (atomic mass unit) is appropriate.

The volume of a nucleus was considered to be negligible compared to the volume of an atom.. The binding energy of a nucleus is the energy holding a nucleus together.. Nucleus occupies an extremely small volume inside the atom.

Atomic structure name draw five protons in the nucleus of the atom. What element is represented by the diagram? Atomic structure fundamentals learning objectives to review the basics concepts of atomic structure that … But dalton or amu (atomic mass unit) is appropriate. Label them with their charge. According to his atom diagram, the atom has a small, positively charged nucleus in center. Draw five protons in the nucleus of the atom.

Draw three electron§ in the second energy level and label them with their charge. Atomic basics answer key part a: What element is represented by the diagram? The binding energy of a nucleus is the energy holding a nucleus together. The atomic number determines the atom's identity electronic cloud nucleus a simplified view of the hydrogen atom, which consists of only one electron outside the nucleus. Draw five protons in the nucleus of the atom. Label them with their charge. The numbers of subatomic particles in an atom can be calculated from its atomic number and mass number. Label them with their charge.
